CEFALY® ENHANCED
Product Code: CEFALYENHANCED
CEFALY® ENHANCED is the first FDA / TGA cleared medical device of its kind for treatment of migraines and is CE marked. The device uses electric impulses that are delivered to the main branch of the trigeminal nerve to prevent and treat pain in the head area. This method can replace or considerably reduce the consumption of painkilling medication and helps to find a better quality of life.
Introducing CEFALY® ENHANCED for Migraine Treatment
Following evidence gathered over the years, several recent technical advances have been made to the CEFALY® device for migraine relief. We now introduce CEFALY® ENHANCED, a new version which is much more efficient than previous models.
Studies, including a recent ACME study, have demonstrated the efficacy and safety of using the new CEFALY® ENHANCED device. Research findings have found the device an effective acute treatment for migraine attacks to relieve headache pain.
The device contains 2 settings:
- Setting 1: an improved acute mode (now 1 hour per session)
- Setting 2: the existing prevention mode (remains at 20 minutes per session)
The previous 3rd setting (sedative effect) has been removed. Both setting 1 and 2 have the sedative effects, allowing the device to focus on managing migraine pain.

Long Term Patient Compliance
Collected data on 14,745 migraine patients who use the CEFALY® ENHANCED device for more than 3 months (between 4 and 19 months) reveals a long-term compliance of 72.4%; much better than for oral preventative migraine medication.
60 Day Cooling-Off Period
If within 60 days, you find that you did not see benefit from CEFALY® ENHANCED please return the device to BHR in Nuneaton. Only products returned in their entirety and including the device, case and charger and in their original packaging can be accepted for return. Items missing will not be refunded. (All items are disposed of using correct disposal procedures as per our Standard Operating Procedures for environmental disposal of electrical items) This option applies only to online sales to consumers.
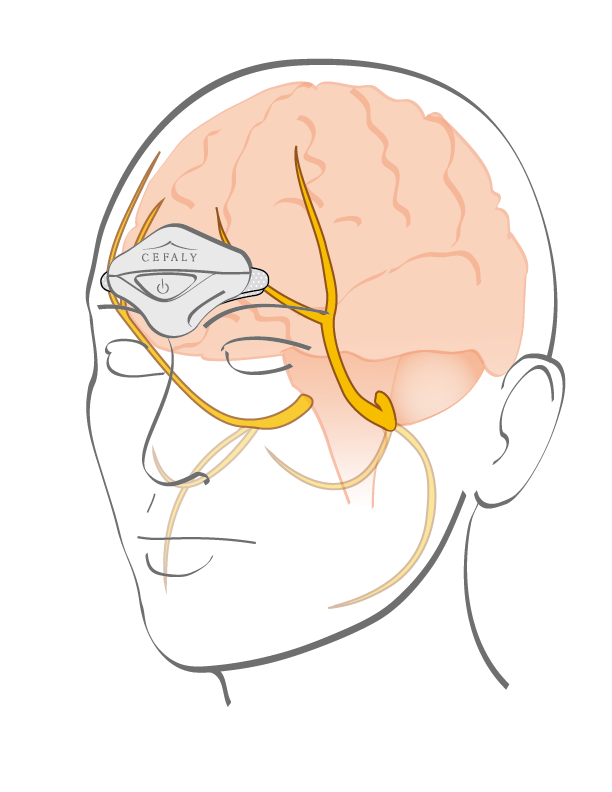
How to use the CEFALY® ENHANCED
Electrode care and maintenance
Face washing tips
The set includes
- CEFALY® ENHANCED
- 3 Reuseable Electrodes
- Resealable Electrode Storage Bag
- Instruction Manual
- Charging Dock & USB Cable
- Storage Case
Each electrode can be used multiple times. Additional packs can be purchased directly from BHR.
User Guide
Clinical Studies
- Supraorbital transcutaneous neurostimulation has sedative effects in healthy subjects
- External Trigeminal Nerve Stimulation for the Acute Treatment of Migraine: Open-Label Trial on Safety and Efficacy.
- A survey on migraine attack treatment with the CEFALY® device in regular users
- Abortive Treatment of Migraine with the Cefaly® Abortive Program Device
- Migraine prevention with a supraorbital transcutaneous stimulator
- Safety and patients’ satisfaction of transcutaneous Supraorbital NeuroStimulation (tSNS) with the Cefaly® device in headache treatment: a survey of 2,313 headache sufferers in the general population
- Transcutaneous supraorbital neurostimulation in “de novo” patients with migraine without aura: the first Italian experience
- Transcutaneous supraorbital neurostimulation for the prevention of chronic migraine: a prospective, open-label preliminary trial
- Clinical experience with transcutaneous supraorbital nerve stimulation in patients with refractory migraine or with migraine and intolerance to topiramate: a prospective exploratory clinical study
- Cerebral metabolism before and after external trigeminal nerve stimulation in episodic migraine
- Migraine treatment with external trigeminal nerve stimulation: current knowledge on
- mechanisms
- Functional Changes of the Perigenual Part of the Anterior Cingulate Cortex after External Trigeminal Neurostimulation in Migraine Patients
- Transcutaneous Supraorbital Nerve Stimulation (t-SNS) with the Cefaly® Device for Migraine Prevention: A Review of the Available Data
- Effects of external trigeminal nerve stimulation (eTNS) on laser evoked cortical potentials (LEP): A pilot study in migraine patients and controls.
- Rationale for electrical parameter determination in external trigeminal nerve stimulation (eTNS) for migraine: A narrative review.







